Boron trifluoride etherate 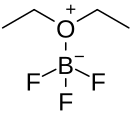 |
 |
| Names |
| Other names Boron Trifluoride Ethyl Ether
Boron Trifluoride Diethyl Etherate |
| Identifiers |
CAS Number | |
3D model (JSmol) | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.355  |
| | |
| UNII | |
| UN number | 2604 |
| | - DTXSID90985377, DTXSID601015537 DTXSID40861733, DTXSID90985377, DTXSID601015537

|
InChI=1S/C4H10O.BF3/c1-3-5-4-2;2-1(3)4/h3-4H2,1-2H3; Key: KZMGYPLQYOPHEL-UHFFFAOYSA-N
|
| |
| Properties |
Chemical formula | C4H10BF3O |
| Molar mass | 141.93 g·mol−1 |
| Appearance | colorless liquid |
| Density | 1.15 g cm3 |
| Melting point | −58 °C (−72 °F; 215 K) |
| Boiling point | 126 °C (259 °F; 399 K) |
| Hazards |
| Occupational safety and health (OHS/OSH): |
Main hazards | Flammable, Reacts with water, Corrosive |
| GHS labelling: |
Pictograms |      |
| | Danger |
| NFPA 704 (fire diamond) | |
| Flash point | 58.5 °C (137.3 °F; 331.6 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references |
Chemical compound
Boron trifluoride etherate, strictly boron trifluoride diethyl etherate, or boron trifluoride–ether complex, is the chemical compound with the formula BF3O(C2H5)2, often abbreviated BF3OEt2. It is a colorless liquid, although older samples can appear brown. The compound is used as a source of boron trifluoride in many chemical reactions that require a Lewis acid.[1] The compound features tetrahedral boron coordinated to a diethylether ligand.[2] Many analogues are known, including the methanol complex.
Reactions
Boron trifluoride etherate serves as a source of boron trifluoride according to the equilibrium:
- BF3OEt2
 BF3 + OEt2
BF3 + OEt2
The BF3 binds to even weak Lewis bases, inducing reactions of the resulting adducts with nucleophiles.[1]
References
- ^ a b Veronica Cornel; Carl J. Lovely (2007). "Boron Trifluoride Etherate". Encyclopedia of Reagents for Organic Synthesis. John Wiley & Sons. pp. rb249.pub2. doi:10.1002/047084289X.rb249.pub2. ISBN 978-0-471-93623-7.
- ^ V. V. Saraev; P. B. Kraikivskii; I. Svoboda; A. S. Kuzakov; R. F. Jordan (2008). "Synthesis, Molecular Structure, and EPR Analysis of the Three-Coordinate Ni(I) Complex [Ni(PPh3)3][BF4]". J. Phys. Chem. A. 112 (48): 12449–12455. Bibcode:2008JPCA..11212449S. doi:10.1021/jp802462x. PMID 18991433.
| Boron pnictogenides | |
|---|
| Boron halides | - BBr3
- BCl3
- BF
- BFO
- BF3
- BI3
- B2F4
- B2Cl4
|
|---|
| Acids | |
|---|
| Boranes | - BH3
- B2H4
- B2H6
- BH3NH3
- B4H10
- B5H9
- B5H11
- B6H10
- B6H12
- B10H14
- B18H22
|
|---|
| Boron oxides and sulfides | |
|---|
| Carbides | |
|---|
| Organoboron compounds | - (BH2Me)2
- BMe3
- BEt3
- Ac4(BO3)2
- COBH3
|
|---|
|
|---|
Salts and covalent derivatives of the fluoride ion |
|---|
| HF | | | | ?HeF2 | | LiF | BeF2 | BF
BF3
B2F4
+BO3 | CF4
CxFy
+CO3 | NF3
FN3
N2F2
NF
N2F4
NF2
?NF5 | OF2
O2F2
OF
O3F2
O4F2
?OF4 | F2 | Ne | | NaF | MgF2 | AlF
AlF3 | SiF4 | P2F4
PF3
PF5 | S2F2
SF2
S2F4
SF3
SF4
S2F10
SF6
+SO4 | ClF
ClF3
ClF5 | ?ArF2
?ArF4 | | KF | CaF
CaF2 | | ScF3 | TiF2
TiF3
TiF4 | VF2
VF3
VF4
VF5 | CrF2
CrF3
CrF4
CrF5
?CrF6 | MnF2
MnF3
MnF4
?MnF5 | FeF2
FeF3
FeF4 | CoF2
CoF3
CoF4 | NiF2
NiF3
NiF4 | CuF
CuF2
?CuF3 | ZnF2 | GaF2
GaF3 | GeF2
GeF4 | AsF3
AsF5 | Se2F2
SeF4
SeF6
+SeO3 | BrF
BrF3
BrF5 | KrF2
?KrF4
?KrF6 | | RbF | SrF
SrF2 | | YF3 | ZrF2
ZrF3
ZrF4 | NbF4
NbF5 | MoF4
MoF5
MoF6 | TcF4
TcF
5
TcF6 | RuF3
RuF
4
RuF5
RuF6 | RhF3
RhF4
RhF5
RhF6 | PdF2
Pd[PdF6]
PdF4
?PdF6 | Ag2F
AgF
AgF2
AgF3 | CdF2 | InF
InF3 | SnF2
SnF4 | SbF3
SbF5 | TeF4
?Te2F10
TeF6
+TeO3 | IF
IF3
IF5
IF7
+IO3 | XeF2
XeF4
XeF6
?XeF8 | | CsF | BaF2 | | LuF3 | HfF4 | TaF5 | WF4
WF5
WF6 | ReF4
ReF5
ReF6
ReF7 | OsF4
OsF5
OsF6
?OsF
7
?OsF
8 | IrF2
IrF3
IrF4
IrF5
IrF6 | PtF2
Pt[PtF6]
PtF4
PtF5
PtF6 | AuF
AuF3
Au2F10
?AuF6
AuF5•F2 | Hg2F2
HgF2
?HgF4 | TlF
TlF3 | PbF2
PbF4 | BiF3
BiF5 | ?PoF2
PoF4
PoF6 | AtF
?AtF3
?AtF5 | RnF2
?RnF
4
?RnF
6 | | FrF | RaF2 | | LrF3 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | | | ↓ | | | | | LaF3 | CeF3
CeF4 | PrF3
PrF4 | NdF2
NdF3
NdF4 | PmF3 | SmF2
SmF3 | EuF2
EuF3 | GdF3 | TbF3
TbF4 | DyF2
DyF3
DyF4 | HoF3 | ErF3 | TmF2
TmF3 | YbF2
YbF3 | | AcF3 | ThF3
ThF4 | PaF4
PaF5 | UF3
UF4
UF5
UF6 | NpF3
NpF4
NpF5
NpF6 | PuF3
PuF4
PuF5
PuF6 | AmF2
AmF3
AmF4
?AmF6 | CmF3
CmF4
?CmF6 | BkF3
BkF
4 | CfF3
CfF4 | EsF3
EsF4
?EsF6 | Fm | Md | No | |
|
| PF6−, AsF6−, SbF6− compounds | - AgPF6
- KAsF6
- LiAsF6
- NaAsF6
- HPF6
- HSbF6
- NH4PF6
- LiSbF6
- KPF6
- KSbF6
- LiPF6
- NaPF6
- NaSbF6
- TlPF6
|
|---|
| AlF6− compounds | - (NH4)3[AlF6]
- Cs2AlF5
- Li3AlF6
- K3AlF6
- Na3AlF6
|
|---|
chlorides, bromides, iodides
and pseudohalogenides | |
|---|
| SiF62-, GeF62- compounds | - BaSiF6
- BaGeF6
- (NH4)2SiF6
- Na2[SiF6]
- K2[SiF6]
- Li2GeF6
- Li2SiF6
|
|---|
| Oxyfluorides | - BrOF3
- BrO2F
- BrO3F
- LaOF
- ThOF2
- VOF
3 - TcO
3F - WOF
4 - YOF
- ClOF3
- ClO2F3
|
|---|
| Organofluorides | - CBrF3
- CBr2F2
- CBr3F
- CClF3
- CCl2F2
- CCl3F
- CF2O
- CF3I
- CHF3
- CH2F2
- CH3F
- C2Cl3F3
- C2H3F
- C6H5F
- C7H5F3
- C15F33N
- C3H5F
- C6H11F
|
|---|
with transition metal,
lanthanide, actinide, ammonium | - VOF3
- CrOF4
- CrF2O2
- NH4F
- (NH4)3CrF6
- (NH4)3GaF6
- (NH4)2GeF6
- (NH4)3FeF6
- (NH4)3InF6
- NH4NbF6
- (NH4)2SnF6
- NH4TaF6
- (NH4)3VF6
- (NH4)2ZrF6
- CsXeF7
- Li2SnF6
- Li2TiF6
- LiWF6
- Li2ZrF6
- K2TiF6
- Rb2TiF6
- Na2TiF6
- Na2ZrF6
- K2NbF7
- K2TaF7
- K2ZrF6
- UO2F2
|
|---|
| nitric acids | |
|---|
| bifluorides | |
|---|
thionyl, phosphoryl,
and iodosyl | - F2OS
- F3OP
- PSF3
- IOF3
- IO3F
- IOF5
- IO2F
- IO2F3
|
|---|
|
Authority control databases: National  | |
|---|




















